f3SMA3
ICGi003-A
General
Donor Information
General Donor Information |
|
| Sex | female |
Phenotype and Disease related information (Donor) |
|
| Diseases | A disease was diagnosed.
|
Karyotyping (Donor) |
|
| Has the donor karyotype been analysed? |
Unknown
|
Other Genotyping (Donor) |
|
| Is there genome-wide genotyping or functional data available? |
No
|
External Databases (Donor) |
|
| BioSamples | SAMEA6877530 |
Ethics
| Has informed consent been obtained from the donor of the embryo/tissue from which the pluripotent stem cells have been derived? | Yes |
| Was the consent voluntarily given? | Yes |
| Has the donor been informed that participation will not directly influence their personal treatment? | Yes |
| Can you provide us with a copy of the Donor Information Sheet provided to the donor? | Yes |
| Do you (Depositor/Provider) hold the original Donor Consent Form? | Yes |
| Please indicate whether the data associated with the donated material has been pseudonymised or anonymised. | pseudonymised |
| Does consent explicitly allow the derivation of pluripotent stem cells? | Yes |
| Does consent prevent CELLS DERIVED FROM THE DONATED BIOSAMPLE from being made available to researchers anywhere in the world? | No |
| How may genetic information associated with the cell line be accessed? | Controlled Access |
| Will the donor expect to receive financial benefit, beyond reasonable expenses, in return for donating the biosample? | No |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the Research Protocol including the consent provisions? | Yes |
| Name of accrediting authority involved? | the D.O. Ott Research Institute of Obstetrics, Gynecology and Reproductology ethics committee |
| Approval number | 65/2014 |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the PROPOSED PROJECT, involving use of donated embryo/tissue or derived cells? | Yes |
| Name of accrediting authority involved? | the D.O. Ott Research Institute of Obstetrics, Gynecology and Reproductology ethics committee |
| Approval number | 65/2014 |
| For generation of the cell line, who was the supplier of any recombined DNA vectors or commercial kits used? |
hIPSC Derivation
General |
|
| Source cell line name | f3SMA |
| Source cell type |
A leukocyte with a single non-segmented nucleus in the mature form.
Synonyms
|
| Source cell origin |
Body substance which consists of plasma and blood cells
Synonyms
|
| Passage number reprogrammed | 6 |
Reprogramming method |
|
| Vector type | Non-integrating |
| Vector | Episomal |
| Genes | |
| Is reprogramming vector detectable? |
No |
| Methods used |
PCR
|
| Files and images showing reprogramming vector expressed or silenced | |
Vector free reprogramming |
|
Other |
|
| Selection criteria for clones | ES-like morphology |
| Derived under xeno-free conditions |
No |
| Derived under GMP? |
No |
| Available as clinical grade? |
No |
Culture Conditions
| Surface coating | Gelatin | |||||||||||||||||||||
| Feeder cells |
mouse embryonic fibroblast cell Cellfinder Ont Id: EFO_0004040 |
|||||||||||||||||||||
| Passage method |
Enzymatically
TrypLE
|
|||||||||||||||||||||
| CO2 Concentration | 5 % | |||||||||||||||||||||
| Medium |
Other medium:
Base medium: KO-DMEM
Main protein source: Knock-out serum replacement Serum concentration: 15 % Supplements
|
|||||||||||||||||||||
| Has Rock inhibitor (Y27632) been used at passage previously with this cell line? | Yes |
|||||||||||||||||||||
| Has Rock inhibitor (Y27632) been used at cryo previously with this cell line? | Yes |
|||||||||||||||||||||
| Has Rock inhibitor (Y27632) been used at thaw previously with this cell line? | Yes |
Characterisation
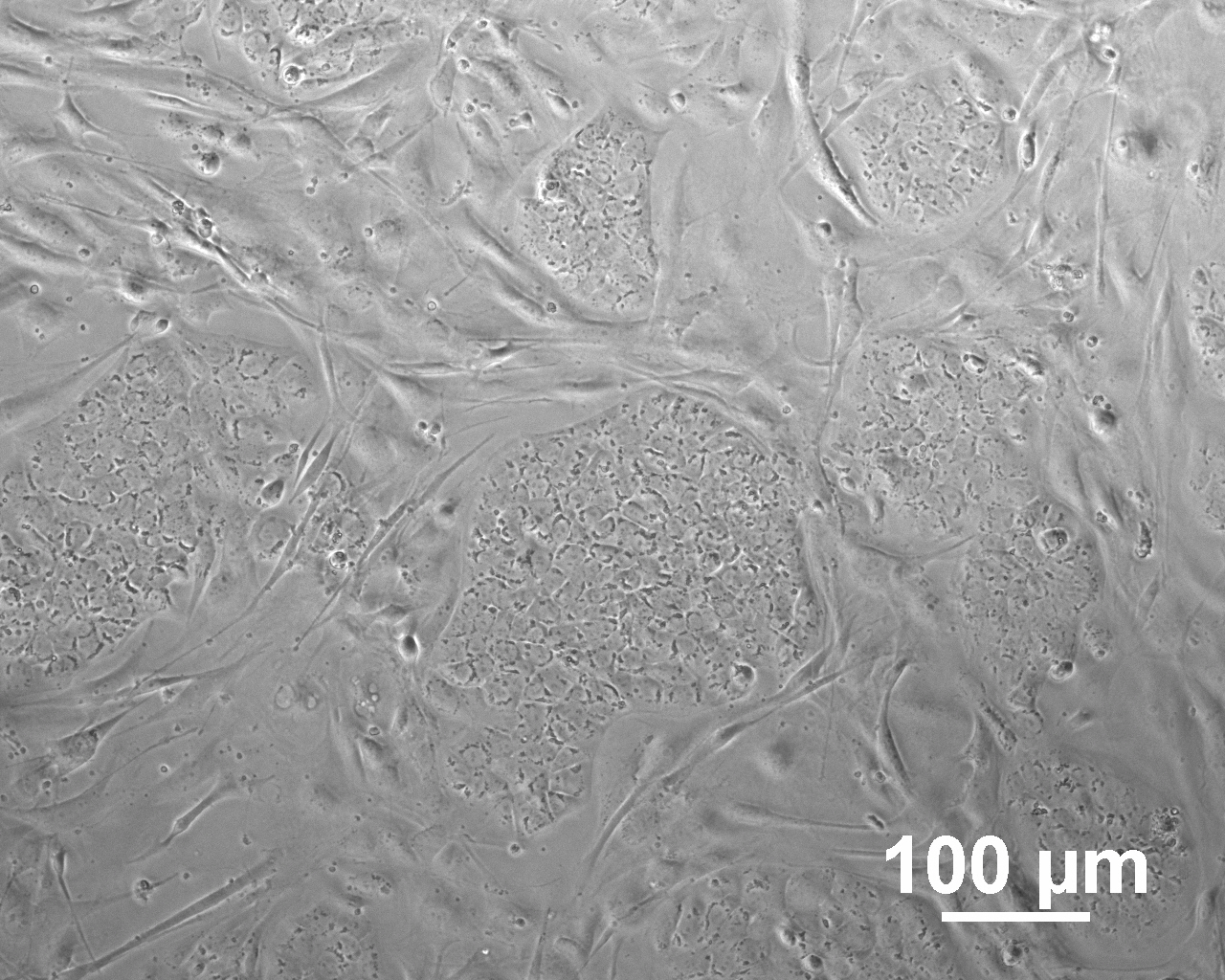
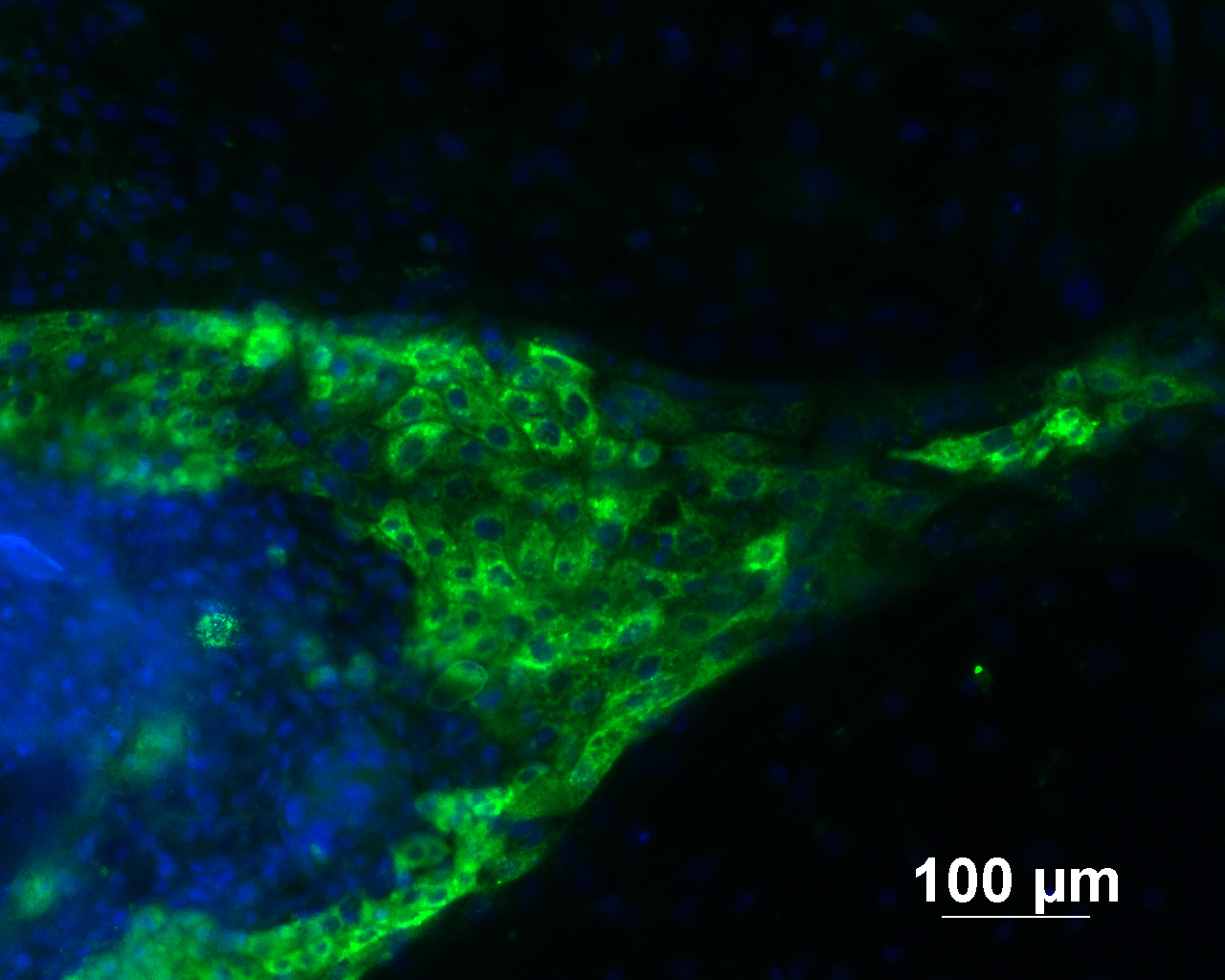
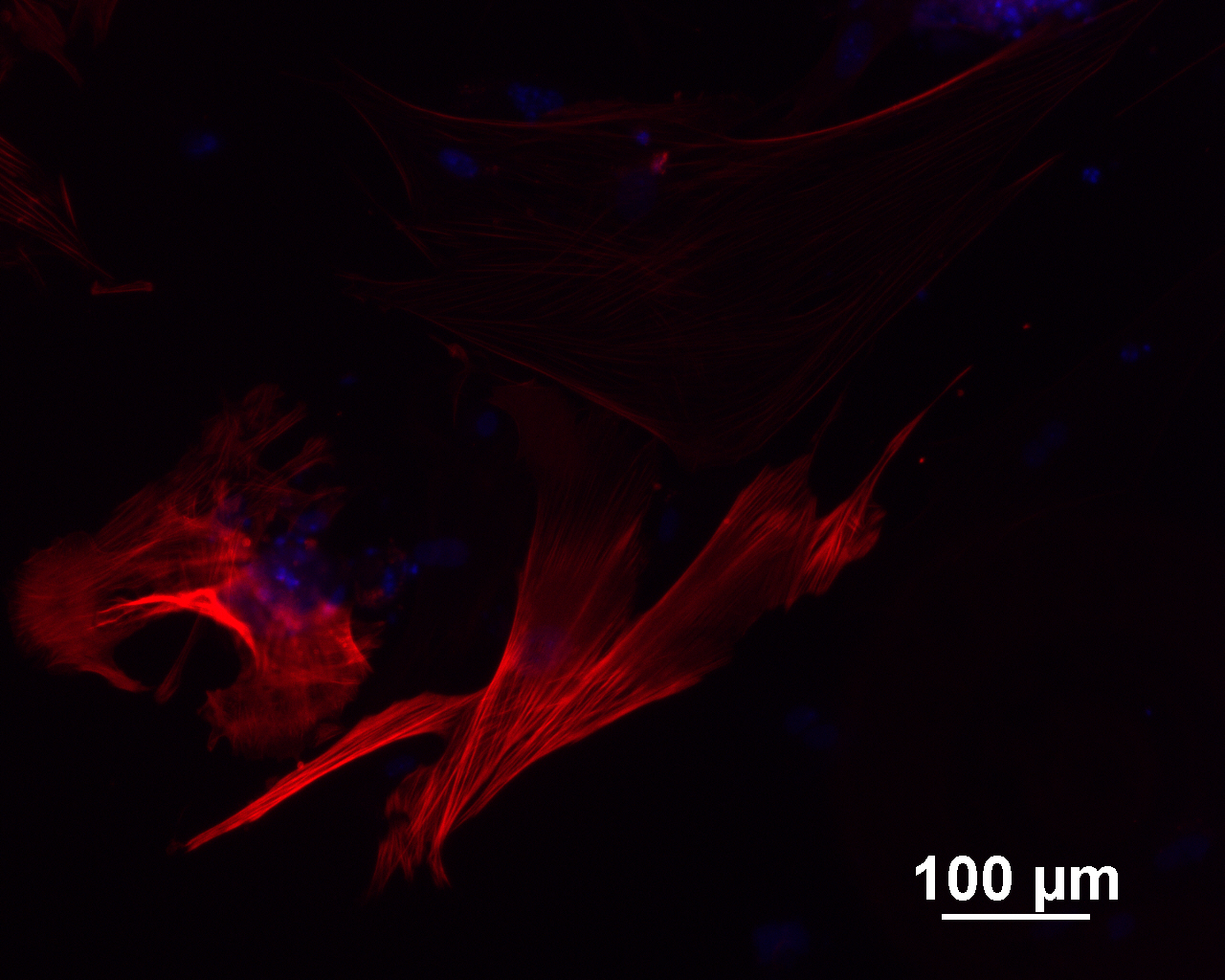
Analysis of Undifferentiated Cells
| Marker | Expressed | Immunostaining | RT-PCR | Flow Cytometry | Enzymatic Assay | Expression Profiles |
| POU5F1 (OCT-4) |
Yes |
|||||
| NANOG |
Yes |
|
||||
| SOX2 |
Yes |
|
||||
| SSEA-4 |
Yes |
|||||
| TRA 1-60 |
Yes |
Score:
| Marker | Present | Absent |
| mCpG | ||
| OCT4 |
Differentiation Potency
In vitro spontaneous differentiation
| Marker | Expressed |
| AFP |
Yes |
In vitro spontaneous differentiation
| Marker | Expressed |
| NEFH |
Unknown |
Microbiology / Virus Screening |
|
| Mycoplasma | Negative |
Genotyping
Karyotyping (Cell Line) |
|
| Has the cell line karyotype been analysed? |
Yes
|
Other Genotyping (Cell Line) |
|



Login to share your feedback, experiences or results with the research community.