NP0081-11
UKKi019-B
General
Cell Line |
|
| hPSCreg name | UKKi019-B |
| Cite as: | UKKi019-B (RRID:CVCL_LD29) |
| Alternative name(s) |
NP0081-11
|
| Cell line type | Human induced pluripotent stem cell (hiPSC) |
| Similar lines |
UKKi034-C (NP0079-C, NP0079-16H) Donor's gene variants: KCNQ1, KCNQ1 Donor diseases: Congenital long QT syndrome UKKi034-A (NP0079-A, NP0079-7B) Donor's gene variants: KCNQ1, KCNQ1 Donor diseases: Congenital long QT syndrome UKKi034-B (NP0079-B, NP0079-15B) Donor's gene variants: KCNQ1, KCNQ1 Donor diseases: Congenital long QT syndrome |
| Last update | 18th October 2019 |
| User feedback | |
Provider |
|
| Generator | Klinikum der Universität zu Köln (UKK) |
| Owner | Institute for Neurophysiology, Medical Faculty |
| Distributors | |
| Derivation country | Germany |
External Databases |
|
| BioSamples | SAMEA17626168 |
| Cellosaurus | CVCL_LD29 |
| Wikidata | Q54990470 |
General Information |
|
| Projects | |
| * Is the cell line readily obtainable for third parties? |
Yes Research use: allowed
Clinical use: allowed
Commercial use: allowed
|
Donor Information
General Donor Information |
|
| Sex | female |
| Age of donor (at collection) | 20-24 |
| Ethnicity | caucasian / european |
Phenotype and Disease related information (Donor) |
|
| Diseases | A disease was diagnosed.
|
| Is the medical history available upon request? | yes |
| Is clinical information available? | yes |
Karyotyping (Donor) |
|
| Has the donor karyotype been analysed? |
No
|
Other Genotyping (Donor) |
|
| Is there genome-wide genotyping or functional data available? |
No
|
Donor Relations |
|
| Other cell lines of this donor | |
External Databases (Donor) |
|
| BioSamples | SAMEA17625418 |
Ethics
| Has informed consent been obtained from the donor of the embryo/tissue from which the pluripotent stem cells have been derived? | Yes |
| Was the consent voluntarily given? | Yes |
| Has the donor been informed that participation will not directly influence their personal treatment? | Yes |
| Can you provide us with a copy of the Donor Information Sheet provided to the donor? | Yes |
| Do you (Depositor/Provider) hold the original Donor Consent Form? | No |
| If you do not hold the Donor Consent Form, do you know who does? | Yes |
| Is there other documentation provided to the donor for consenting purposes? | No |
| Confirm that consent was obtained by a qualified professional | Yes |
| Has the donor agreed to be re-contacted? | Yes |
| Has the donor been informed about how her/his data will be protected? | Yes |
| Please indicate whether the data associated with the donated material has been pseudonymised or anonymised. | pseudonymised |
| Does consent explicitly allow the derivation of pluripotent stem cells? | Yes |
| * Does consent expressly prevent the derivation of pluripotent stem cells? | No |
| * Does consent pertain to a specific research project? | No |
| Does consent permit unforeseen future research, without further consent? | Yes |
| Does the consent permit uses of donated embryo/tissue or derived cell line intended for clinical treatment or human applications? | No |
| Does consent expressly prevent development of commercial products? | No |
| Does consent expressly prevent financial gain from any use of the donated embryo/tissue, including any product made from it? | No |
| Does consent expressly permit storage of donated embryo/tissue for an unlimited time? | Yes |
| Does consent expressly permit storage of cells derived from the donated embryo/tissue for an unlimited time? | Yes |
| Does consent prevent the DONATED BIOSAMPLE from being made available to researchers anywhere in the world? | No |
| Does consent prevent CELLS DERIVED FROM THE DONATED BIOSAMPLE from being made available to researchers anywhere in the world? | No |
Does consent permit research by | |
| an academic institution? | Yes |
| a public organisation? | Yes |
| a non-profit company? | Yes |
| a for-profit corporation? | Yes |
| Does consent expressly permit collection of genetic information? | Yes |
| Does consent expressly permit storage of genetic information? | Yes |
| Does consent prevent dissemination of genetic information? | No |
| Has the donor been informed that their donated biosample or derived cells may be tested for the presence of microbiological agents / pathogens? | Yes |
| Has the donor consented to receive information discovered during use of donated embryo/tissue that has significant health implications for the donor? | No |
| How may genetic information associated with the cell line be accessed? | Controlled Access |
| Will the donor expect to receive financial benefit, beyond reasonable expenses, in return for donating the biosample? | No |
| Does the consent anticipate that the donor will be notified of results or outcomes of any research involving the donated samples or derived cells? | No |
| Does the consent permit the donor, upon withdrawal of consent, to stop the use of the derived cell line(s) that have already been created from donated samples? | No |
| Does the consent permit the donor, upon withdrawal of consent, to stop delivery or use of information and data about the donor? | No |
| Does consent permit access to medical records of the donor? | Yes |
| Please describe how access is provided: | Clinic in which the donor was recruted holds the medical records |
| Does consent permit access to any other source of information about the clinical treatment or health of the donor? | Yes |
| Contact data, institution, or website: | |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the Research Protocol including the consent provisions? | Yes |
| Name of accrediting authority involved? | Independent Ethics Committee of the Faculty of Medicine of the University of Cologne |
| Approval number | DRKS00009433 |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the PROPOSED PROJECT, involving use of donated embryo/tissue or derived cells? | Yes |
| Name of accrediting authority involved? | Ethics Committee of the Faculty of Medicine of the University of Cologne |
| Approval number | DRKS00009433 |
| Do you have obligations to third parties in regard to the use of the cell line? | No |
| Are you aware of any further constraints on the use of the donated embryo/tissue or derived cells? | No |
| For generation of the cell line, who was the supplier of any recombined DNA vectors or commercial kits used? | Thermo Fisher, Cytotune iPS 2.0 Sendai reprogramming kit |
| Are you aware of any constraints on the use or distribution of the cell line from the owner or any parties identified in the query above? | Yes |
| Constraints for use or distribution | Commercial use of this cell line is not permitted |
hIPSC Derivation
General |
|
| Source cell type |
A leukocyte with a single non-segmented nucleus in the mature form found in the circulatory pool of blood.
Synonyms
|
| Age of donor (at collection) | 20-24 |
| Collected in | 2016 |
| Passage number reprogrammed | 0 |
Reprogramming method |
|
| Vector type | Non-integrating |
| Vector | Sendai virus |
| Genes | |
| Is reprogramming vector detectable? |
No |
| Methods used |
Immunostaining, PCR
|
| Notes on reprogramming vector detection | sendai virus absent |
| Files and images showing reprogramming vector expressed or silenced | |
Vector free reprogramming |
|
| Type of used vector free reprogramming factor(s) |
None
|
Other |
|
| Selection criteria for clones | morphology |
| Derived under xeno-free conditions |
Yes |
| Derived under GMP? |
No |
| Available as clinical grade? |
No |
Culture Conditions
| Surface coating | Vitronectin | |||||||||
| Feeder cells |
No |
|||||||||
| Passage method |
Enzyme-free cell dissociation
EDTA
|
|||||||||
| O2 Concentration | 20 % | |||||||||
| CO2 Concentration | 5 % | |||||||||
| Medium |
Essential 8™
Supplements
|
|||||||||
| Has Rock inhibitor (Y27632) been used at passage previously with this cell line? | No |
|||||||||
| Has Rock inhibitor (Y27632) been used at cryo previously with this cell line? | No |
|||||||||
| Has Rock inhibitor (Y27632) been used at thaw previously with this cell line? | No |
Characterisation
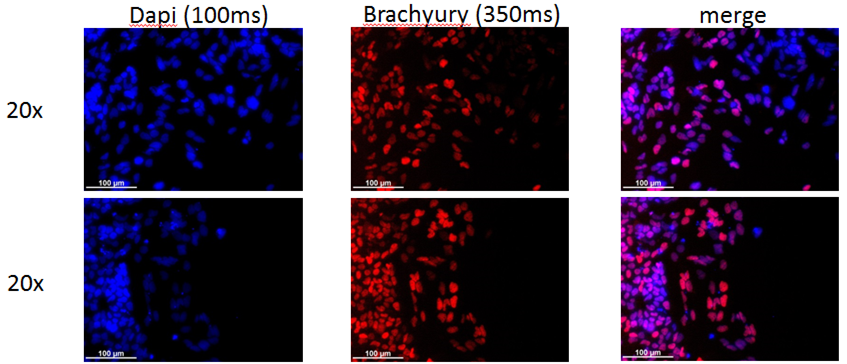
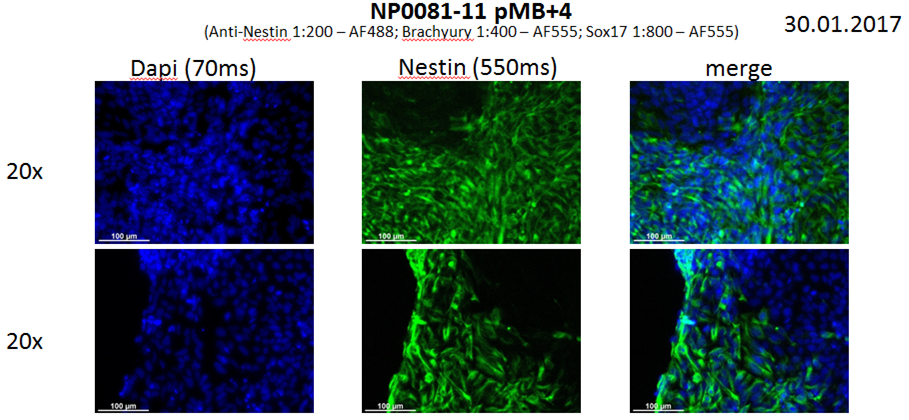
Analysis of Undifferentiated Cells
Differentiation Potency
Microbiology / Virus Screening |
|
| HIV 1 | Negative |
| HIV 2 | Negative |
| Hepatitis B | Negative |
| Hepatitis C | Negative |
| Mycoplasma | Negative |
Certificate of Analysis |
|
| Is there a certificate of analysis available? |
Yes
Passage:
41
|
Genotyping
Karyotyping (Cell Line) |
|
| Has the cell line karyotype been analysed? |
Yes
No larger chromosomal aberrations to be reported
Passage number: 38
Karyotyping method:
Molecular karyotyping by SNP array
|
Other Genotyping (Cell Line) |
|
| Is there genome-wide genotyping or functional data available? |
Yes
Whole genome sequencing
https://ega-archive.org/studies/EGAS00001002755 This cell line has undergone WGS using the Illumina HiSeq X platform at 30x coverage. Fastq files are stored at the European Genome Archive, users can apply for access to this data by submitting an application form to the EBiSC Data Access Committee https://ega-archive.org/dacs/EGAC00001000768 |


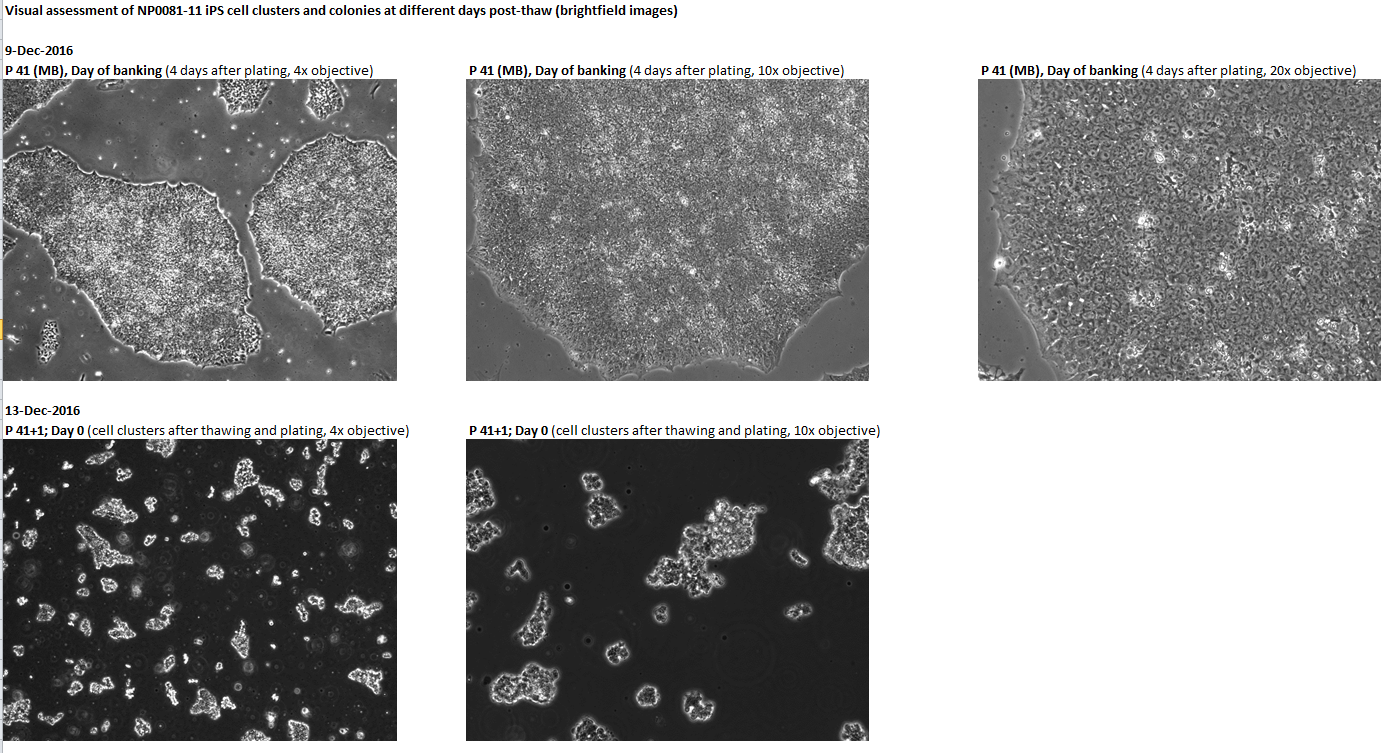
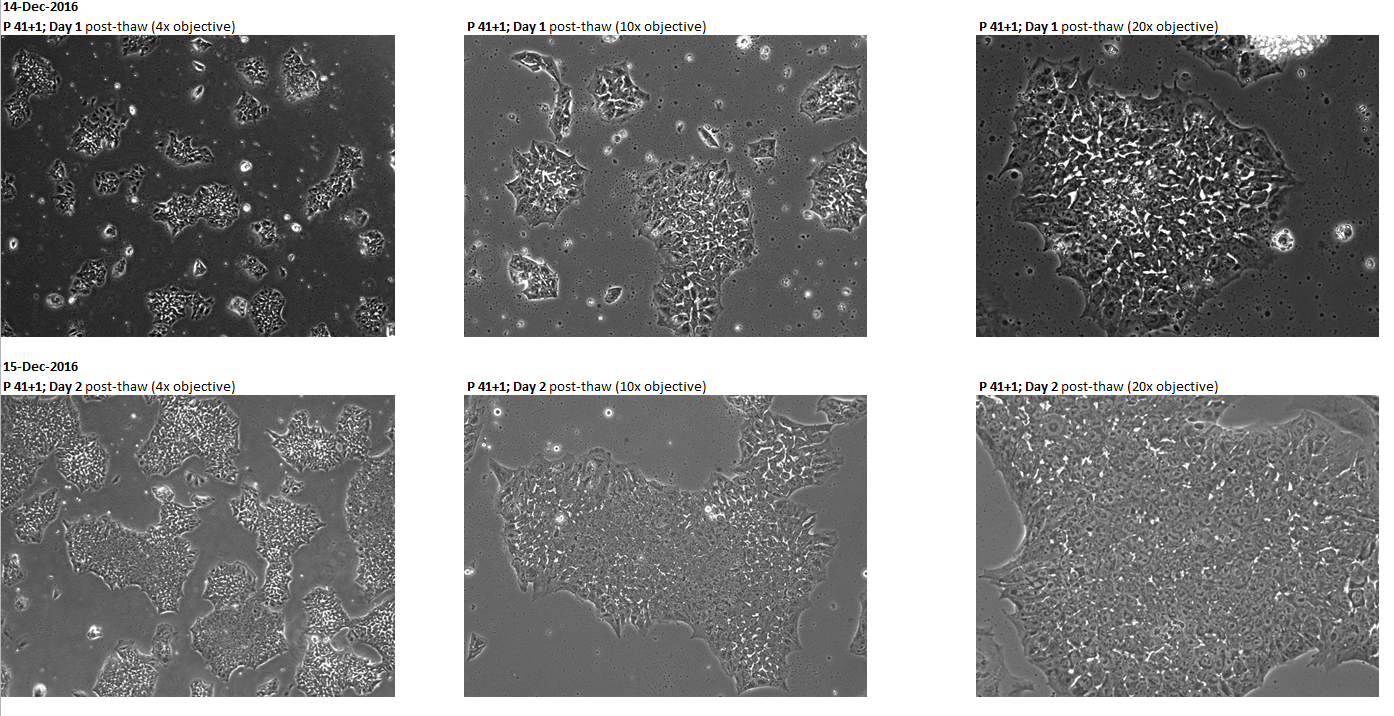
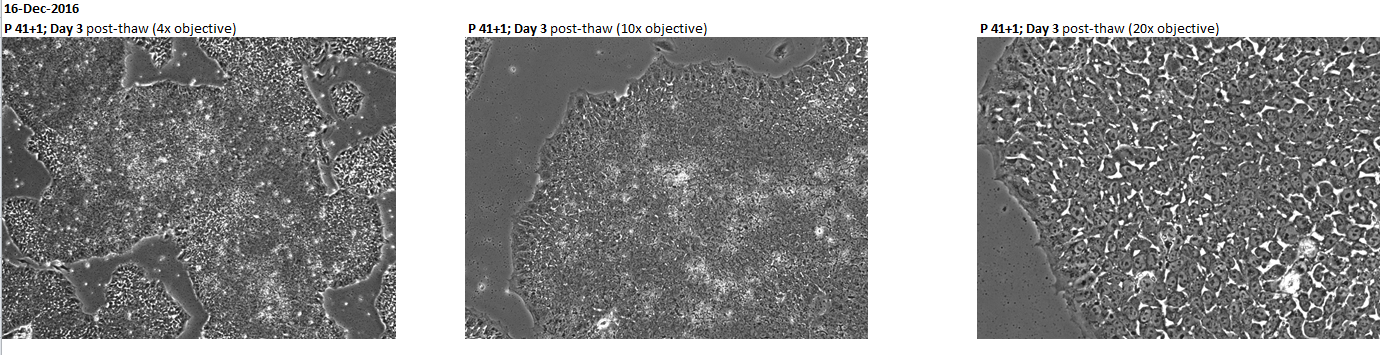
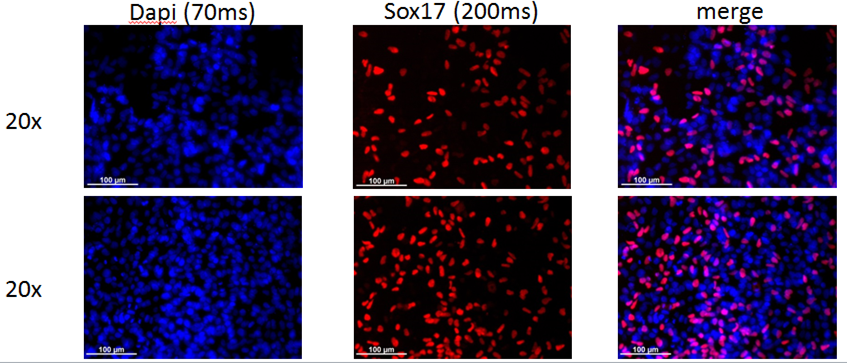
Login to share your feedback, experiences or results with the research community.