Phx_SRCAP_g3_1400_6/8
MHHi001-A-14
General
Cell Line |
|
| hPSCreg name | MHHi001-A-14 |
| Cite as: | MHHi001-A-14 |
| Alternative name(s) |
Phx_SRCAP_g3_1400_6/8
|
| Cell line type | Human induced pluripotent stem cell (hiPSC) |
| Similar lines |
MHHi001-A-16 (PHX_CRISPR_JR_DIS10) MHHi001-A-17 (PHX_CRISPR_JR_DIS13) MHHi001-A-13 (Phx_SRCAP_g3_1200_7) MHHi001-A-12 (Phx_SRCAP_g3_1200_4) MHHi001-A (hHSC_Iso4_ADCF_SeViPS2 (Phönix)) UMGWi004-B-1 (BNH1_iPSC_FHS1_cr_well7_JR) UMGWi004-B-2 (BNH1_iPSC_FHS1_cr_well12_JR) MHHi001-A-7 (NRF2_A_Phönix-iPSC clone19) MHHi001-A-20 (cyclinB1-eGFP hiPSCs) MHHi001-A-6 (NRF2_A_Phönix-iPSC clone12) |
| Last update | 11th July 2025 |
| User feedback | |
Provider |
|
| Generator | Department of Functional Genomics - Human Molecular Genetics (UMGW) |
External Databases |
|
| BioSamples | SAMEA114222274 |
General Information |
|
| Publications | |
| * Is the cell line readily obtainable for third parties? |
Yes Research use: allowed
Clinical use: not allowed
Commercial use: not allowed
|
| Subclone of | |
Genetic Modification
| Disease/phenotype related modifications |
|
Donor Information
General Donor Information |
|
| Sex | female |
| Ethnicity | Caucasian |
Phenotype and Disease related information (Donor) |
|
| Diseases | No disease was diagnosed.
|
| Family history | N/A |
| Is the medical history available upon request? | no |
| Is clinical information available? | no |
Other Genotyping (Donor) |
|
| Is there genome-wide genotyping or functional data available? |
No
|
External Databases (Donor) |
|
| BioSamples | SAMEA4564585 |
Ethics
Also have a look at the ethics information for the parental line
MHHi001-A
.
| For generation of the cell line, who was the supplier of any recombined DNA vectors or commercial kits used? |
hIPSC Derivation
General |
|
|
The source cell information can be found in the parental cell line
MHHi001-A.
|
|
Reprogramming method |
|
| Vector type | Non-integrating |
| Vector | Sendai virus |
| Genes | |
| Is reprogramming vector detectable? |
No |
| Methods used |
RT-PCR
|
| Files and images showing reprogramming vector expressed or silenced | |
Vector free reprogramming |
|
Other |
|
| Selection criteria for clones | Sanger sequencing for mutations in target region Frameshift analysed using online tools TIDE (https://tide.nki.nl/) and indigo (https://www.gear-genomics.com/indigo/) |
| Derived under xeno-free conditions |
No |
| Derived under GMP? |
No |
| Available as clinical grade? |
No |
Culture Conditions
| Surface coating | Matrigel/Geltrex | ||||||
| Feeder cells |
No |
||||||
| Passage method | Mechanically | ||||||
| CO2 Concentration | 5 % | ||||||
| Medium |
mTeSR™ 1
Supplements
|
||||||
| Has Rock inhibitor (Y27632) been used at passage previously with this cell line? | No |
||||||
| Has Rock inhibitor (Y27632) been used at cryo previously with this cell line? | No |
||||||
| Has Rock inhibitor (Y27632) been used at thaw previously with this cell line? | Yes |
Characterisation
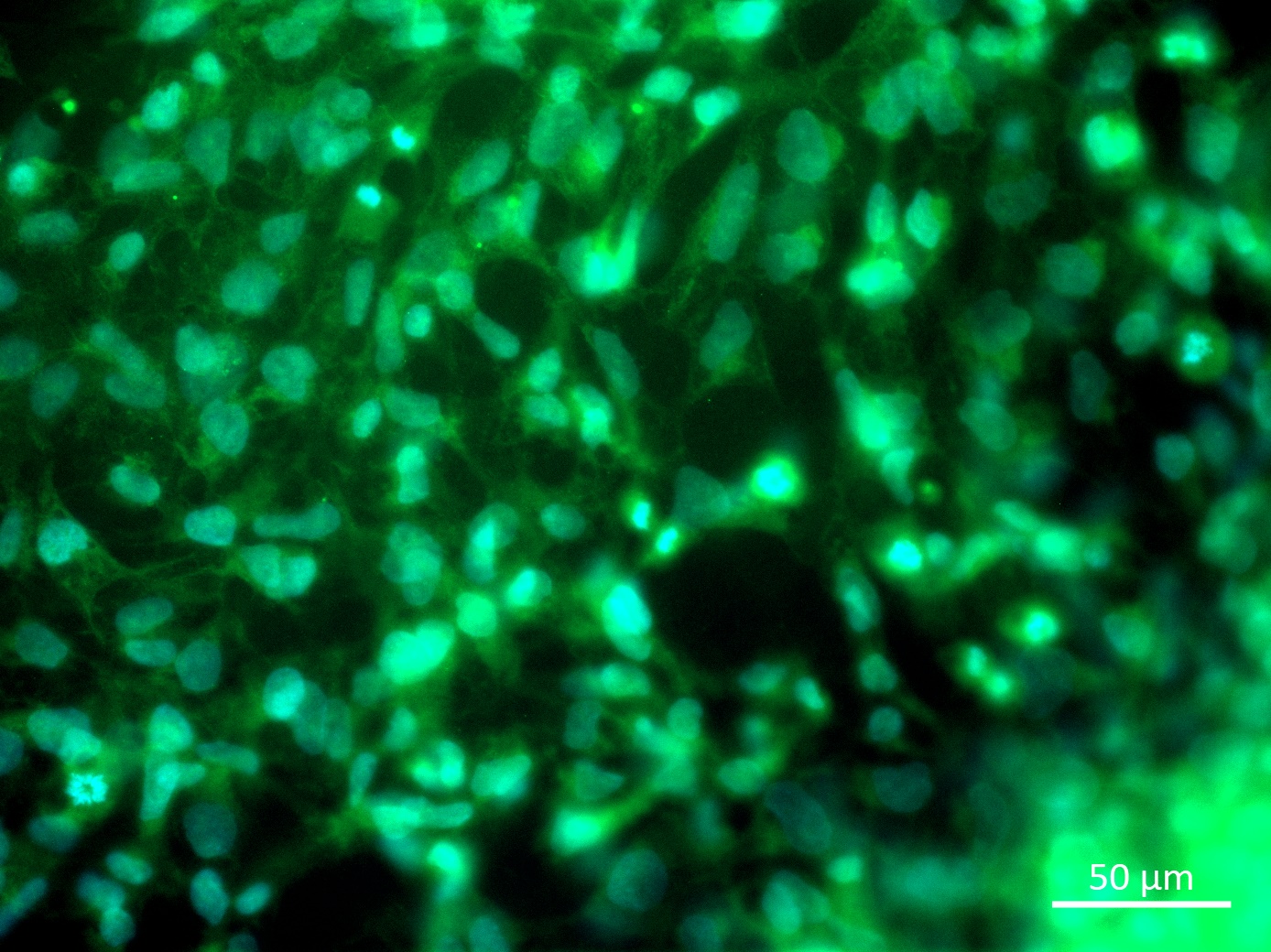
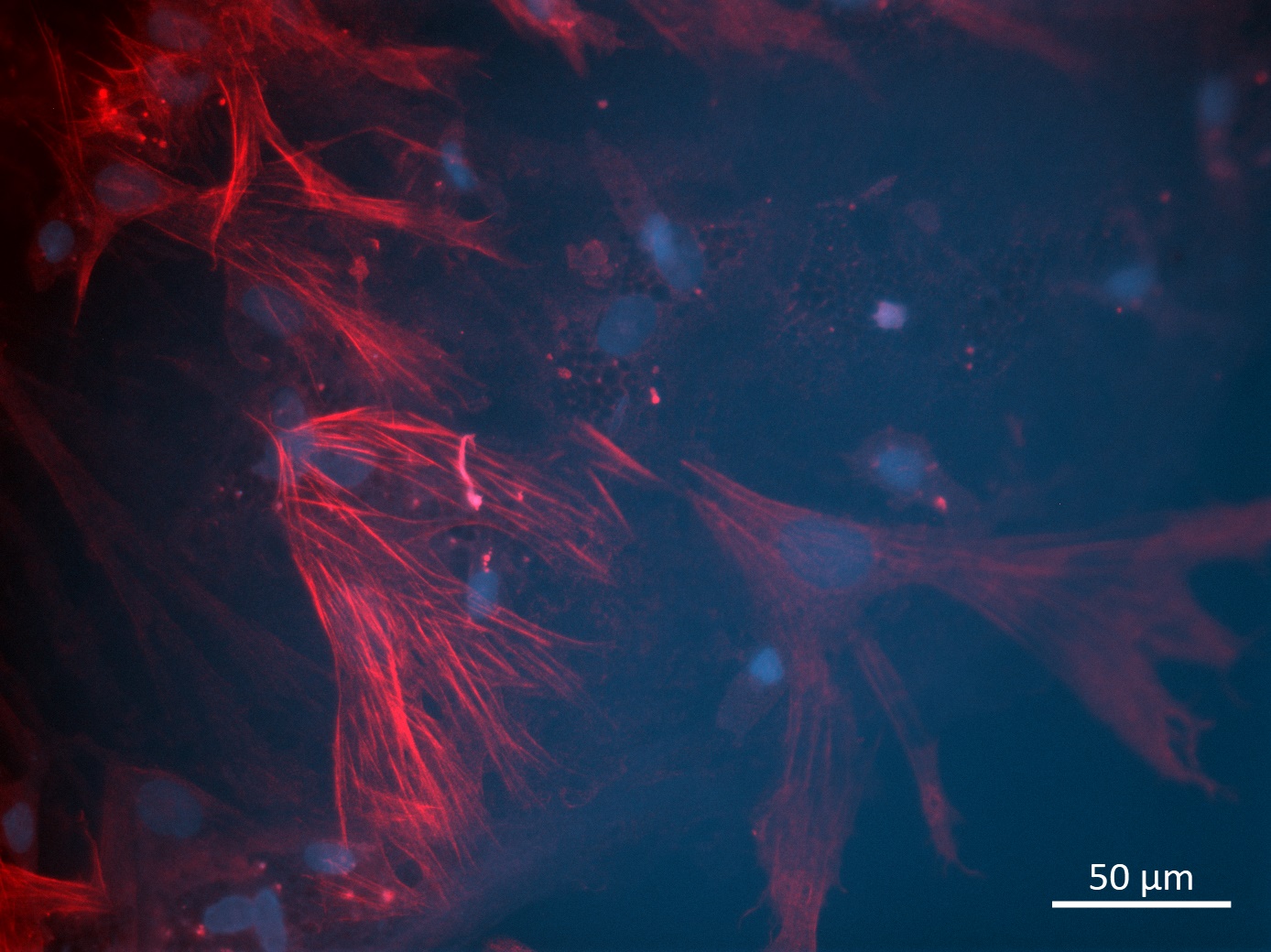
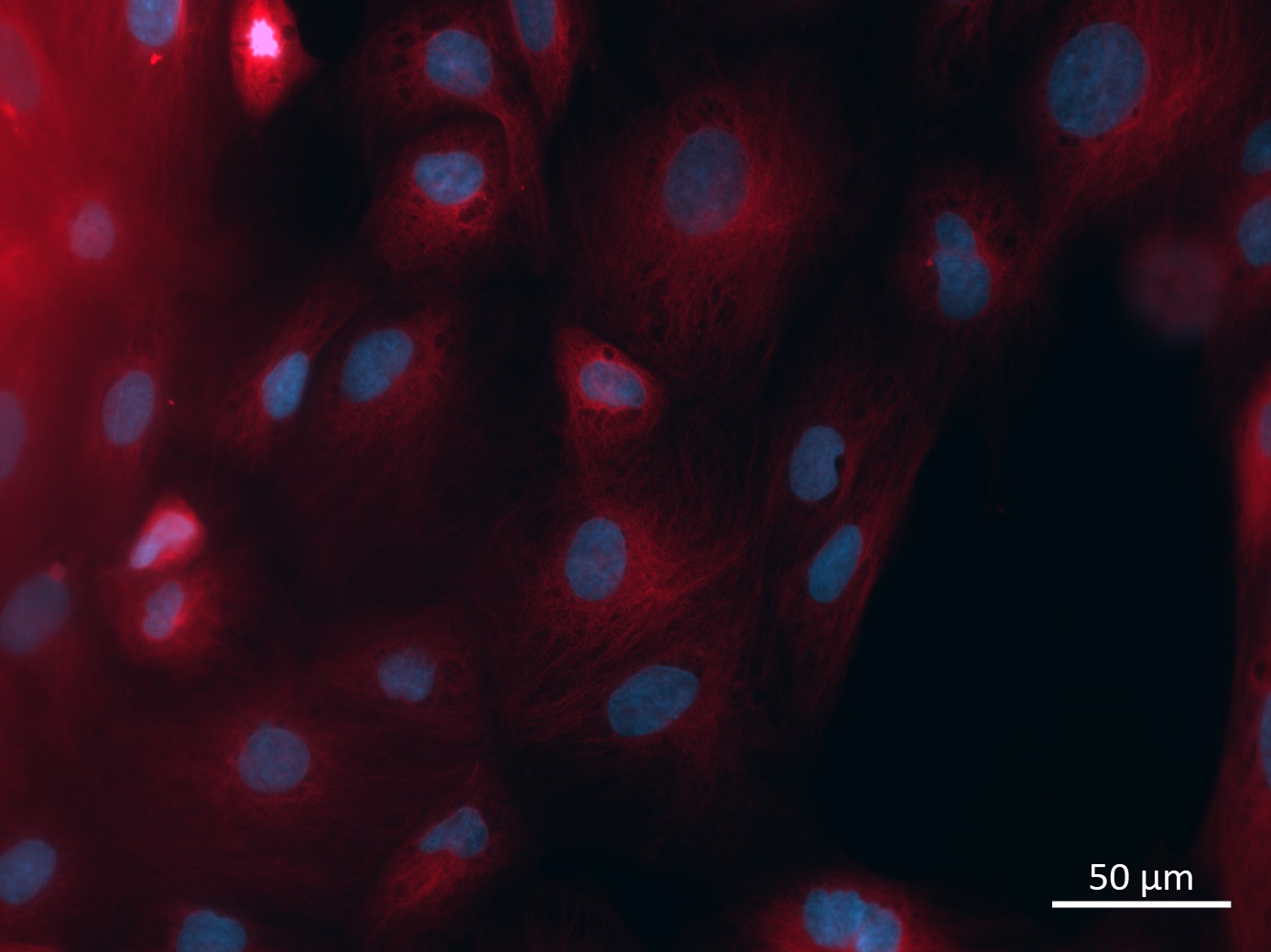
Analysis of Undifferentiated Cells
| Marker | Expressed | Immunostaining | RT-PCR | Flow Cytometry | Enzymatic Assay | Expression Profiles |
| NANOG |
Yes |
|||||
| POU5F1 (OCT-4) |
Yes |
|||||
| SOX2 |
Yes |
|||||
| TRA 1-60 |
Yes |
Differentiation Potency
In vitro spontaneous differentiation
Protocol or reference
In vitro spontaneous differentiation
Protocol or reference
Microbiology / Virus Screening |
|
| Mycoplasma | Negative |
Genotyping
Karyotyping (Cell Line) |
|
| Has the cell line karyotype been analysed? |
Yes
46,XX
Passage number: 10
Karyotyping method:
low coverage whole genome sequencing on NextSeq 550
|
Other Genotyping (Cell Line) |
|


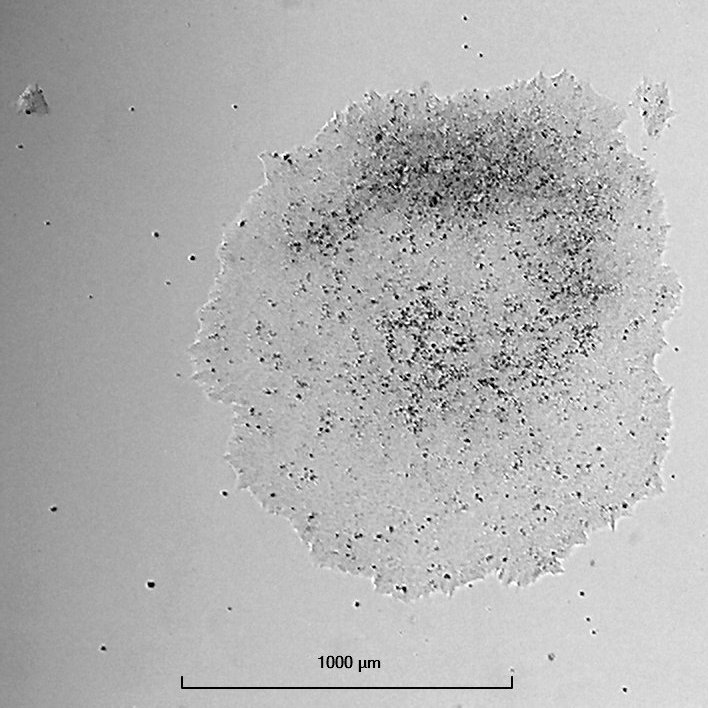
Login to share your feedback, experiences or results with the research community.