OF0176-EYS02-C7, EYS02-MiPS4F7
CABi002-A
General
iPSC Line |
|
| hPSCreg name | CABi002-A |
| Cite as: | CABi002-A (RRID:CVCL_UJ72) |
| Alternative name(s) |
OF0176-EYS02-C7, EYS02-MiPS4F7
|
| iPSC line type | Human induced pluripotent stem cell (hiPSC) |
| Similar iPSC lines |
LUMCi056-A-1 (LUMC0128iCRB01 heterozygote CRISPR corrected isogenic clone 02, iso02LUMC0128iCRB01) Donor's gene variants: CRB1, CRB1 Donor diseases: Retinitis Pigmentosa ESi077-A (CABi001-A, PRPF31-MiPS4F3) Donor's gene variants: PRPF31, PRPF31 Donor diseases: Retinitis Pigmentosa LUMCi055-A (CRB1 patient 117 compound heterozygous 2983G>T p.(Glu995*) c.1892A>G, p.(Tyr631Cys), LUMC0117iCRB01) Donor's gene variants: CRB1, CRB1 Donor diseases: Retinitis Pigmentosa LUMCi056-A (CRB1 patient 128 compound heterozygous c.2843G>A p.(Cys948Tyr) and c.3122T>C p.(Met1041Thr), LUMC0128iCRB01) Donor's gene variants: CRB1, CRB1 Donor diseases: Retinitis Pigmentosa |
| Last update | 30th March 2021 |
| User feedback | |
Provider |
|
| Generator | CABIMER (Andalusian Molecular Biology and Regenerative Medicine Centre) (CAB) |
External iPSC Databases |
|
| Cellosaurus | CVCL_UJ72 |
| BioSamples | SAMEA8268542 |
| Wikidata | Q93441237 |
General iPSC Information |
|
| Publications | |
| * Is the cell line readily obtainable for third parties? |
No |
Donor Information
General Donor Information |
|
| Sex | male |
Phenotype and Disease related information (Donor) |
|
| Diseases | A disease was diagnosed.
|
External Databases (Donor) |
|
| BioSamples | SAMEA8268543 |
Ethics
| Has informed consent been obtained from the donor of the embryo/tissue from which the pluripotent stem cells have been derived? | Yes |
| Was the consent voluntarily given? | Yes |
| Has the donor been informed that participation will not directly influence their personal treatment? | Yes |
| Can you provide us with a copy of the Donor Information Sheet provided to the donor? | Yes |
| Do you (Depositor/Provider) hold the original Donor Consent Form? | Yes |
| Please indicate whether the data associated with the donated material has been pseudonymised or anonymised. | pseudonymised |
| Does consent explicitly allow the derivation of pluripotent stem cells? | Yes |
| Does consent prevent CELLS DERIVED FROM THE DONATED BIOSAMPLE from being made available to researchers anywhere in the world? | No |
| How may genetic information associated with the cell line be accessed? | Controlled Access |
| Will the donor expect to receive financial benefit, beyond reasonable expenses, in return for donating the biosample? | No |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the Research Protocol including the consent provisions? | Yes |
| Name of accrediting authority involved? | Comité Coordinador de Ética de a Investigación Biomédica de Andalucía |
| Approval number | PR-01-2015 |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the PROPOSED PROJECT, involving use of donated embryo/tissue or derived cells? | Yes |
| Name of accrediting authority involved? | Comité Coordinador de Ética de a Investigación Biomédica de Andalucía |
| Approval number | PR-01-2015 |
| For generation of the cell line, who was the supplier of any recombined DNA vectors or commercial kits used? |
iPSC Derivation
General |
|
| Source cell type |
A peripheral blood cell with a single nucleus. This category includes lymphocytes and monocytes.
Synonyms
|
| Source cell origin |
The portion of the upper extremity between the shoulder and the elbow. For clinical purposes this term is also used to refer to the whole superior limb.
Synonyms
|
Reprogramming method |
|
| Vector type | Non-integrating |
| Vector | Sendai virus |
| Is reprogramming vector detectable? |
No |
| Methods used |
RT-PCR
|
| Files and images showing reprogramming vector expressed or silenced | |
Vector free reprogramming |
|
Other |
|
| Derived under xeno-free conditions |
Unknown |
| Derived under GMP? |
Unknown |
| Available as clinical grade? |
Unknown |
iPSC Culture Conditions
| Medium |
mTeSR™ 1
|
iPSC Characterisation
Analysis of undifferentiated iPSCs
Differentiation Potency
Genotyping
Karyotyping (iPSC Line) |
|
| Has the iPSC line karyotype been analysed? |
Yes
|
Other Genotyping (iPSC Line) |
|


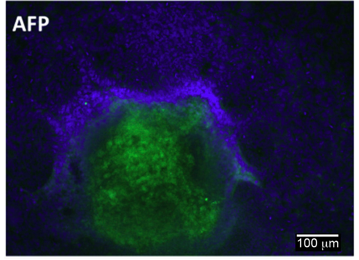
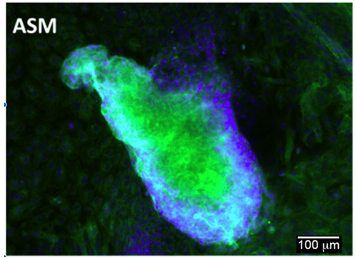
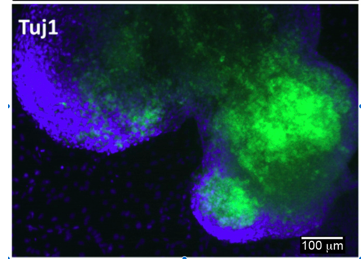
Login to share your feedback, experiences or results with the research community.