m55Alz-15L
ICGi008-B
General
Donor Information
General Donor Information |
|
| Sex | male |
| Age of donor (at collection) | 55-59 |
| Ethnicity | Caucasian |
Phenotype and Disease related information (Donor) |
|
| Diseases | A disease was diagnosed.
|
Karyotyping (Donor) |
|
| Has the donor karyotype been analysed? |
No
|
Other Genotyping (Donor) |
|
| Is there genome-wide genotyping or functional data available? |
No
|
Donor Relations |
|
| Other cell lines of this donor | |
External Databases (Donor) |
|
| BioSamples | SAMEA5341577 |
Ethics
| Has informed consent been obtained from the donor of the embryo/tissue from which the pluripotent stem cells have been derived? | Yes |
| Was the consent voluntarily given? | Yes |
| Has the donor been informed that participation will not directly influence their personal treatment? | Yes |
| Can you provide us with a copy of the Donor Information Sheet provided to the donor? | Yes |
| Do you (Depositor/Provider) hold the original Donor Consent Form? | Yes |
| Alternatives to consent are available? | No |
| Is there other documentation provided to the donor for consenting purposes? | No |
| Confirm that consent was obtained by a qualified professional | Yes |
| Has the donor agreed to be re-contacted? | Unknown |
| Has the donor been informed about how her/his data will be protected? | Yes |
| Please indicate whether the data associated with the donated material has been pseudonymised or anonymised. | pseudonymised |
| Does consent explicitly allow the derivation of pluripotent stem cells? | Yes |
| * Does consent expressly prevent the derivation of pluripotent stem cells? | No |
| * Does consent pertain to a specific research project? | No |
| Does consent permit unforeseen future research, without further consent? | Yes |
| Does the consent permit uses of donated embryo/tissue or derived cell line intended for clinical treatment or human applications? | No |
| Does consent expressly prevent development of commercial products? | Yes |
| Does consent expressly prevent financial gain from any use of the donated embryo/tissue, including any product made from it? | Yes |
| Does consent expressly permit storage of donated embryo/tissue for an unlimited time? | Yes |
| Does consent expressly permit storage of cells derived from the donated embryo/tissue for an unlimited time? | Yes |
| Does consent prevent the DONATED BIOSAMPLE from being made available to researchers anywhere in the world? | No |
| Does consent prevent CELLS DERIVED FROM THE DONATED BIOSAMPLE from being made available to researchers anywhere in the world? | No |
Does consent permit research by | |
| an academic institution? | Yes |
| a public organisation? | No |
| a non-profit company? | No |
| a for-profit corporation? | No |
| Does consent expressly permit collection of genetic information? | Yes |
| Does consent expressly permit storage of genetic information? | Yes |
| Does consent prevent dissemination of genetic information? | No |
| Has the donor been informed that their donated biosample or derived cells may be tested for the presence of microbiological agents / pathogens? | Yes |
| Has the donor consented to receive information discovered during use of donated embryo/tissue that has significant health implications for the donor? | Yes |
| How may genetic information associated with the cell line be accessed? | Controlled Access |
| Will the donor expect to receive financial benefit, beyond reasonable expenses, in return for donating the biosample? | No |
| Does the consent anticipate that the donor will be notified of results or outcomes of any research involving the donated samples or derived cells? | Yes |
| Does the consent permit the donor, upon withdrawal of consent, to stop the use of the derived cell line(s) that have already been created from donated samples? | Yes |
| Does the consent permit the donor, upon withdrawal of consent, to stop delivery or use of information and data about the donor? | Yes |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the Research Protocol including the consent provisions? | Yes |
| Name of accrediting authority involved? | Center of New Medical Technologies (CNMT) in Akademgorodok |
| Approval number | 21 |
| Has a favourable opinion been obtained from a research ethics committee, or other ethics review panel, in relation to the PROPOSED PROJECT, involving use of donated embryo/tissue or derived cells? | Yes |
| Name of accrediting authority involved? | Center of New Medical Technologies (CNMT) in Akademgorodok |
| Approval number | 21 |
| Do you have obligations to third parties in regard to the use of the cell line? | No |
| Are you aware of any further constraints on the use of the donated embryo/tissue or derived cells? | No |
| Is there an MTA available for the cell line? | No |
| For generation of the cell line, who was the supplier of any recombined DNA vectors or commercial kits used? | Addgene |
| Are you aware of any constraints on the use or distribution of the cell line from the owner or any parties identified in the query above? | No |
hIPSC Derivation
General |
|
| Source cell line name |
m55Alz Derived from same source line (potentially other lot and donor, see below):
|
| Source cell type |
Any skin fibroblast that is part of some dermis.
|
| Source cell origin |
EDITOR_NOTE TODO - distinguish between entire skin of region and arbitrary zone of skin on region; A zone of skin that is part of a back [Automatically generated definition].
Synonyms
|
| Age of donor (at collection) | 55-59 |
| Collected in | 2013 |
| Passage number reprogrammed | 3 |
Reprogramming method |
|
| Vector type | Non-integrating |
| Vector | Episomal |
| Is reprogramming vector detectable? |
No |
| Methods used |
PCR
|
| Files and images showing reprogramming vector expressed or silenced | |
Vector free reprogramming |
|
| Type of used vector free reprogramming factor(s) |
None
|
Other |
|
| Selection criteria for clones | The selection of colonies was carried out according to morphological criteria. We selected flat monolayer colonies with tightly packed cells with high nuclear/cytoplasmic ratio. Embryonic stem cell-like clones were picked by a glass microcapillary. |
| Derived under xeno-free conditions |
No |
| Derived under GMP? |
No |
| Available as clinical grade? |
No |
Culture Conditions
| Surface coating | Gelatin | |||||||||||||||||||||
| Feeder cells |
mouse emryonic fibroblasts |
|||||||||||||||||||||
| Passage method |
Enzymatically
TrypLE
|
|||||||||||||||||||||
| O2 Concentration | 20 % | |||||||||||||||||||||
| CO2 Concentration | 5 % | |||||||||||||||||||||
| Medium |
Other medium:
Base medium: Knockout DMEM (Life Technologies)
Main protein source: Knock-out serum replacement Serum concentration: 15 % Supplements
|
|||||||||||||||||||||
| Has Rock inhibitor (Y27632) been used at passage previously with this cell line? | Yes |
|||||||||||||||||||||
| Has Rock inhibitor (Y27632) been used at cryo previously with this cell line? | No |
|||||||||||||||||||||
| Has Rock inhibitor (Y27632) been used at thaw previously with this cell line? | Yes |
Characterisation
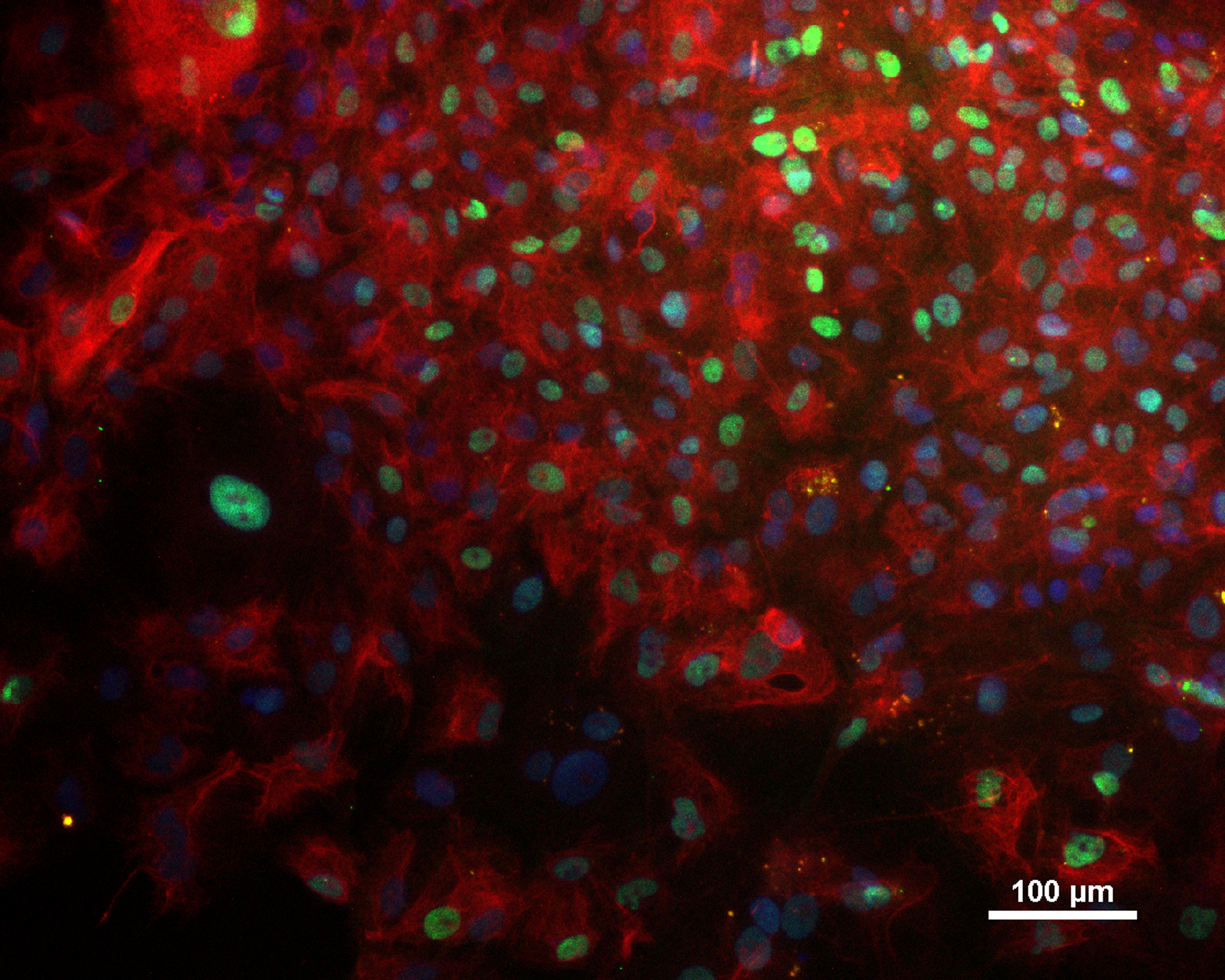
Analysis of Undifferentiated Cells
| Marker | Expressed | Immunostaining | RT-PCR | Flow Cytometry | Enzymatic Assay | Expression Profiles |
| SOX2 |
Yes |
|||||
| NANOG |
Yes |
|||||
| POU5F1 (OCT-4) |
Yes |
|||||
| Alkaline Phosphatase |
Yes |
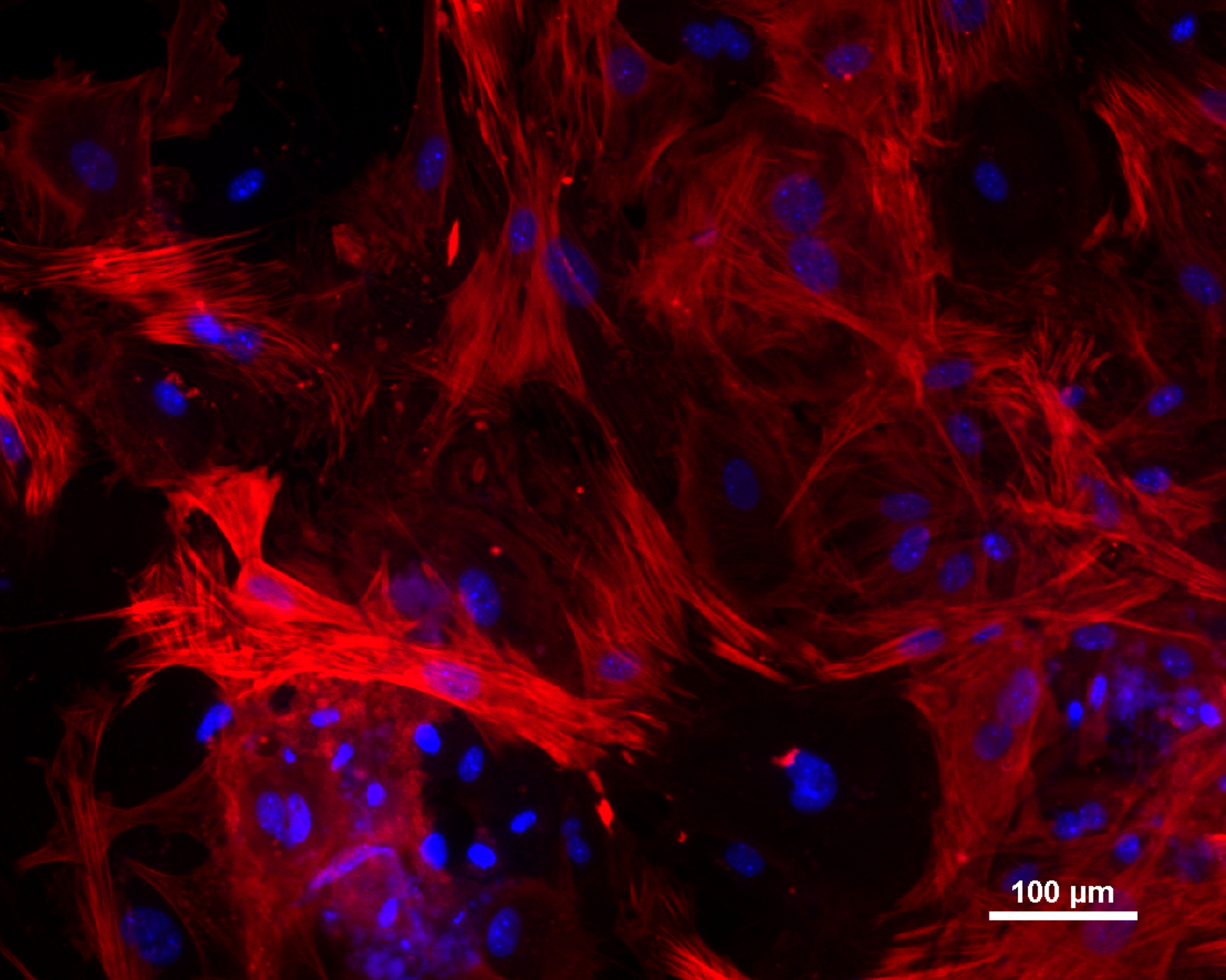
Morphology pictures
m55Alz-15L-1.tif
Morphology of ICGi008-B iPSC, the scale bar - 500 mcm.
Differentiation Potency
In vitro spontaneous differentiation
Microbiology / Virus Screening |
|
| Mycoplasma | Negative |
Genotyping
Karyotyping (Cell Line) |
|
| Has the cell line karyotype been analysed? |
Yes
Karyotyping and G-banding show ICGi008-B iPSC have a normal 46, XX karyotype.
Passage number: 29
Karyotyping method:
G-Banding
|
Other Genotyping (Cell Line) |
|



Login to share your feedback, experiences or results with the research community.